A hygienic-grade flow measurement installation in a modern food and beverage processing facility. Accurate metering is the backbone of product consistency and regulatory compliance.
Evaluating Positive Displacement Flowmeters for Hygienic and High-Viscosity Applications
When a confectionery plant in Germany switched from turbine meters to hygienic positive displacement (PD) flowmeters for measuring glucose syrup, their batch-to-batch weight deviation dropped from ±1.8% to ±0.15% — a 10× improvement that translated directly into a reduction of annual raw material waste worth €120,000. That single instrument change illustrates why the choice of flowmeter technology matters far more than most engineers initially appreciate.
In hygienic and high-viscosity processing — food and beverage, pharmaceutical, biotechnology, personal care — the stakes attached to flow measurement are uniquely high. Contamination from a poorly cleaned meter can trigger a product recall. An inaccurate reading in a pharmaceutical dosing line can constitute a batch failure with regulatory consequences. A meter that stalls on cold chocolate or warm honey can shut down an entire filling station.
This guide examines positive displacement (PD) flowmeters through the lens of those demanding sectors. We cover the physics of how they work, which hygienic standards actually matter and why, how viscosity and temperature interact to affect measurement accuracy, what surface finish figures like Ra 0.8 µm mean in practice, and how to build a defensible business case based on total cost of ownership rather than purchase price alone. Along the way, we share real-world data, decision frameworks, and sector-specific application insights.
Whether you are a process engineer specifying instruments for a new dairy line, a validation specialist qualifying a pharmaceutical filling suite, or a procurement manager evaluating long-term instrument contracts, this resource will give you the technical depth and practical context to make a confident choice.
- Positive Displacement (PD) Flowmeter
- A device that measures fluid flow by repeatedly filling and emptying a precisely defined internal chamber. Every complete cycle counts one fixed volume — no inference from velocity or pressure required.
- Turndown Ratio
- The ratio of maximum to minimum measurable flow rate while maintaining stated accuracy. A 20:1 turndown means the meter stays accurate across a 20-fold range of flow rates.
- CIP / SIP
- Clean-in-Place / Steam-in-Place — automated sanitation methods that clean or sterilise equipment without disassembly, requiring the meter to survive hot caustic solutions or pressurised steam cycles.
- Ra Value
- Arithmetic mean surface roughness in micrometres (µm). Lower Ra = smoother surface = fewer micro-crevices for bacteria or product residue to colonise.
- Repeatability
- The ability of a meter to give the same reading for the same flow condition each time — often more practically important than absolute accuracy in batching and filling operations.
- Slippage (Leakage)
- Internal fluid bypass between the rotating element and the meter housing. Slippage causes under-reading and is reduced by higher fluid viscosity and tighter manufacturing tolerances.
Overview of Positive Displacement Flowmeters
How PD Meters Work and Common Internal Mechanisms
The operating principle of a PD flowmeter is elegantly direct: fluid physically pushes a mechanical element — gears, rotors, a disc, or pistons — around a precisely engineered chamber. Each revolution or stroke displaces a fixed, calibrated volume of liquid. Counting those displacements over time yields volumetric flow rate; integrating them yields totalized volume. Crucially, the meter does not infer flow from velocity, differential pressure, or the fluid’s electrical properties. It counts real volumes.
In hygienic applications, four primary mechanism types dominate:
- Oval Gear Meters — Two elliptical gears mesh and rotate inside a housing. Fluid fills the crescent-shaped cavity between each gear and the housing wall, then is displaced as the gears turn. Each full revolution moves a precise volume four times (two per gear, twice per revolution). They are arguably the most common PD design in hygienic food processing because their smooth internals tolerate CIP well.
- Helical / Bi-Rotor Meters — A pair of intermeshing helical rotors creates a continuous, smooth fluid transfer with very low pulsation. The helical geometry eliminates the stick-slip effect common in spur gear designs, making these meters preferred for shear-sensitive fluids like cream or certain biotech broths.
- Nutating Disk Meters — A disc wobbles (nutates) within a spherical chamber, pushing fluid from inlet to outlet. Each complete nutation cycle equals one fixed volume. Their low internal shear makes them suitable for delicate product streams.
- Reciprocating Piston Meters — One or more pistons stroke back and forth in calibrated cylinders. They excel at very low flow rates (down to a few mL/min) and are frequently found in pharmaceutical dosing and flavour-addition skids.
Video: Positive Displacement Flowmeters — Working Principles, Types, and Industrial Applications (YouTube)
Typical Performance Metrics: Accuracy, Repeatability, and Turndown
The numbers that make PD meters attractive in precision applications are well established by field experience. For viscous fluids above approximately 50 cP, a well-calibrated oval gear or helical rotor meter routinely achieves ±0.2 to ±0.5% of reading (AR) accuracy and repeatability figures as tight as ±0.02 to ±0.05% AR. This repeatability — not absolute accuracy — is often what matters most in batching and filling: if the meter always reads the same way, a single offset correction in the PLC handles the rest.
Turndown ratios of 10:1 to 20:1 are common in hygienic PD meters; some advanced models achieve 50:1 with appropriate electronics. This wide operating window is critical in dairy plants where the same line may run at 20% capacity during startup cleaning cycles and at 100% during peak production.
When PD Meters Are Preferred Over Other Technologies
PD meters outperform alternatives in three specific scenarios: high viscosity (where magnetic and vortex meters either stall or lose accuracy), very low flow rates (where Coriolis meters become cost-prohibitive at small pipe sizes), and situations requiring true volumetric totalization without a secondary calculation from velocity. Turbine meters, by contrast, suffer performance degradation above roughly 500 cP because viscous drag suppresses the rotor; electromagnetic meters simply cannot measure non-conductive fluids like oils, fats, or syrups.
Key Hygienic Design Considerations

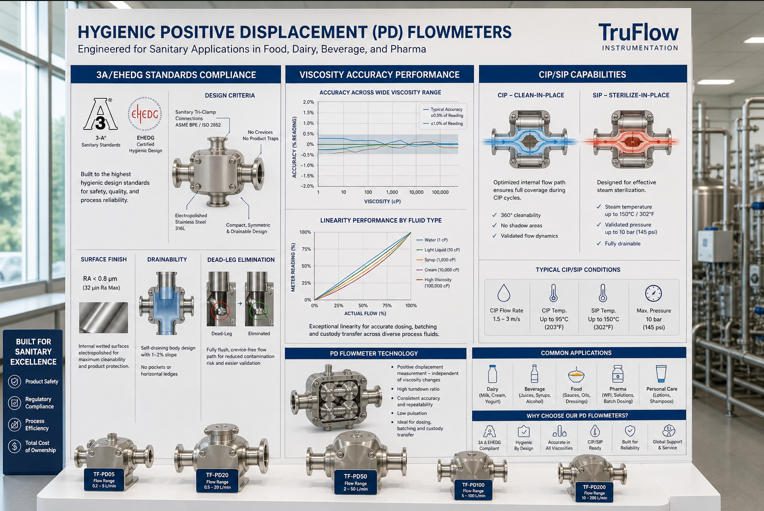
Hygienic-grade PD meters use tri-clamp, RJT, or DIN connections — not threaded fittings — to eliminate thread roots where bacteria can harbour. Surface finish specifications are verified by profilometry before assembly.
Standards and Certifications: 3A, EHEDG, FDA/USDA
Three certification frameworks dominate hygienic flowmeter specification in regulated industries:
| Standard / Body | Region of Influence | Key Requirements for Flowmeters | Relevance to PD Meters |
|---|---|---|---|
| 3-A SSI (3-A Sanitary Standards Inc.) | North America (widely adopted globally) | Minimum Ra 0.8 µm on product-contact surfaces, no dead legs >2× pipe diameter, approved elastomers, cleanable geometry | Mandated on dairy lines in the US; audit trail requires physical 3-A symbol on the instrument |
| EHEDG (European Hygienic Engineering & Design Group) | Europe, Middle East, Asia | Full cleanability validation by authorised test bodies; geometric cleanability maps; thermal resistance to SIP temperatures up to 150 °C | Required on EU dairy, brewery, and pharma lines; validation testing is more rigorous than 3-A self-declaration |
| FDA / USDA | United States (internationally benchmarked) | 21 CFR materials compliance; cGMP alignment; no substances that migrate into food at unsafe levels | All product-contact materials must appear on FDA-accepted lists; relevant to pharmaceutical lines under 21 CFR Part 211 |
Seal Integrity and Cleanability in Sterile Environments
The most common hygienic failure mode in PD flowmeters is not inaccuracy — it is seal degradation. The dynamic seals between rotating elements and the housing are subjected to millions of stress cycles per year. In a pharmaceutical application running at 60 °C with weekly SIP cycles at 130 °C, an elastomer seal that is not FDA-compliant or not rated for thermal cycling will micro-crack and create harbourage points invisible to visual inspection but detectable by environmental monitoring swabs.
EPDM, PTFE-encapsulated, and FFKM (perfluoroelastomer) seals are the materials of choice. PTFE-encapsulated seals are preferred where both FDA compliance and broad chemical resistance are needed simultaneously. For high-temperature SIP applications, FFKM offers the best long-term stability but at higher cost — an appropriate trade-off when a single sterility failure in a biopharmaceutical batch can cost several million dollars.
Mess-Free Disassembly and Inspection for CIP/SIP
A well-designed hygienic PD meter should disassemble in under five minutes with standard sanitary tools — no special spanners, no internal fasteners that trap product, no cavities that retain cleaning solution. The industry benchmark is the “thumb screw” design philosophy: if a maintenance technician cannot fully disassemble, inspect, and reassemble the meter without spilling product or requiring calibrated torque tools, the design is not truly hygienic. Leading manufacturers now publish validated CIP programs specifying flow velocities, temperatures, and chemical concentrations — a level of documentation that regulators increasingly expect to see in hygienic area qualification packages.
Viscosity Compatibility and Measurement Range
Flow Tube Design and Displacement Principles for High Viscosity
The physics of PD metering is uniquely well-suited to viscous fluids. In a magnetic flowmeter, measurement depends on the velocity of a conductive fluid moving through a magnetic field — at very low velocities typical of high-viscosity media, the induced EMF signal becomes too weak to resolve accurately. In a vortex meter, the Strouhal number relationship breaks down below Reynolds numbers of approximately 20,000, which viscous fluids at modest flow rates easily fall below.
A PD meter, by contrast, requires only that the fluid physically push the measuring element around the chamber. Viscosity actually helps — it reduces internal slippage (the thin film of fluid bypassing the measuring element through clearances), thereby improving accuracy at low flow rates. As a practical rule of thumb, PD meter accuracy improves progressively up to approximately 1,000–2,000 cP before it plateaus. Above 10,000 cP, the internal drag torque begins to create measurable pressure drop concerns, but the measurement accuracy itself remains excellent.
±2.0%
±1.5%
±1.0%
±0.5%
±0.2%
~1 cP
(Water)
±2.0%
~50 cP
(Light Oil)
±1.5%
~500 cP
(Glucose)
±0.5%
~2,000 cP
(Honey)
±0.3%
~5,000 cP
(Chocolate)
±0.2%
Fluid Viscosity (cP)
Typical Accuracy (% AR)
PD Meter Accuracy Improves with Higher Viscosity
Illustrative accuracy band for a calibrated oval gear PD meter. At low viscosities, slippage increases measurement uncertainty; higher viscosity reduces slip, tightening the accuracy band. Based on published industry data from multiple manufacturers.
Temperature Effects and Viscosity-Temperature Relationships
Temperature and viscosity are inseparably linked in most product streams, and this relationship has direct consequences for meter performance. Chocolate at 40 °C may have a viscosity of 3,000–5,000 cP; at 30 °C it can exceed 15,000 cP and begin to solidify around internal components at shutdown. Honey at 25 °C sits near 2,000–10,000 cP depending on water content, but warms to 500–1,000 cP at 40 °C. Palm oil transitions from solid at 20 °C to a free-flowing liquid above 35 °C.
Each of these transitions changes the meter’s slippage characteristics, meaning a single calibration curve at one temperature may introduce systematic error at another. Best practice for high-variability products is to perform a multi-point calibration spanning the expected temperature range, or to install an integrated Pt100 temperature sensor and apply a viscosity-temperature correction in the transmitter or DCS. Modern PD transmitters from suppliers such as those linked to by Jade Ant Instruments’ flow measurement comparison guide support this kind of real-time compensation.
Turndown Ratio and Flow Rate Transitions for Viscous Fluids
A turndown ratio of 10:1 is adequate for many continuous-flow applications, but filling and batching operations may demand 20:1 or higher because the flow profile during a batch cycle is inherently variable: a fast fill phase at 80–100% of maximum flow, a dribble phase at 10–15% as the target weight approaches, and a cutoff at near-zero flow. A meter that loses accuracy in the dribble phase introduces systematic overshoot on every batch — a problem that compounds catastrophically at high throughput in a bottling plant running 200 fills per hour.
Materials and Surface Finish for Cleanability
Suitable Materials: Stainless Steel Grades and Elastomers for Hygiene
The gold standard for product-contact surfaces in hygienic PD meters is 316L stainless steel (UNS S31603), a low-carbon austenitic grade that combines corrosion resistance, passivation stability, and weldability. The “L” (low carbon) specification minimises chromium carbide precipitation at weld heat-affected zones — a critical detail because carbide precipitation creates microscopic surface pitting that neither CIP chemicals nor manual polishing can adequately address.
For the housing and non-product-contact structural elements, 304L or 316L is acceptable. Certain meter designs use engineering polymers such as PEEK for the internal rotor — PEEK offers excellent chemical resistance, very low density (reducing bearing loads), and dimensional stability at SIP temperatures up to 150 °C, while eliminating the risk of corrosion between two stainless steel components in the presence of chloride-containing cleaning agents.
Elastomers deserve particular scrutiny. Acceptable materials for most hygienic applications include:
- EPDM — standard choice for hot water, steam, and alkaline CIP; not suitable for petroleum-based products
- FKM (Viton) — good for oils, fats, and mild acids; limited performance above 130 °C in steam
- FFKM — maximum chemical and thermal resistance; specified for pharmaceutical WFI (water for injection) loops and aggressive bio-process streams
- PTFE (or PTFE-encapsulated) — chemically inert, FDA compliant; used as seats and backup seals where dynamic compression sealing is not required
Surface Finish Requirements (Ra Values) and Polish Methods
Surface roughness in hygienic instrumentation is specified by the Ra value — the arithmetic mean of surface profile deviations from the centreline, measured in micrometres. The significance for microbial control is not merely aesthetic: bacteria form biofilms preferentially in surface irregularities. Research by the European Hygienic Engineering & Design Group has demonstrated that Listeria monocytogenes biofilm formation rates on stainless steel surfaces decrease significantly below Ra 0.8 µm and become negligible below Ra 0.4 µm.
| Application | Minimum Ra Requirement | Typical Polish Method | Relevant Standard |
|---|---|---|---|
| General food processing | Ra ≤ 0.8 µm | Mechanical + electropolish | 3-A SSI, EHEDG |
| Dairy / beverage | Ra ≤ 0.8 µm | Electropolish (preferred) | 3-A SSI |
| Pharmaceutical (non-sterile) | Ra ≤ 0.8 µm | Electropolish + passivation | FDA 21 CFR |
| Pharmaceutical (sterile / WFI) | Ra ≤ 0.4 µm | Electropolish + passivation + certification | ASME BPE, FDA |
| Biotech (bioreactor feeds) | Ra ≤ 0.25 µm | Electropolish + passivation + rouge removal | ASME BPE |
Electropolishing — an electrochemical process that removes surface micro-peaks — produces a more uniform, lower Ra finish than mechanical polishing alone and simultaneously enriches the chromium oxide passive layer. This dual effect (smoother surface + better corrosion resistance) explains why ASME BPE (Bioprocessing Equipment) standard specifies electropolish for all sterile-service product-contact surfaces.
Compatibility with Caustics, Acids, and Cleaning Agents
Standard CIP programs in the food industry use 1–2% NaOH at 70–80 °C (alkaline phase) and 0.5–1% nitric or phosphoric acid at 60–70 °C (acid phase). A hygienic PD meter must survive these cycles thousands of times over its service life. 316L stainless steel is compatible with both phases at these concentrations and temperatures. EPDM seals are stable in alkaline CIP but swell in strong nitric acid above 1.5%. FKM seals handle the acid phase but may extract in prolonged alkaline exposure. Knowing the exact CIP chemistry before specifying seals is essential — it is a detail that is frequently overlooked in project specifications until a meter failure in the first six months of operation makes it unmistakable.
Cleaning and Sanitation Protocols
CIP/SIP Integration and Cycle Effectiveness
CIP (Clean-in-Place) effectiveness in a PD meter depends on three physical factors: turbulent flow velocity through the meter body (minimum 1.5 m/s is the industry target for dairy lines), dwell time at cleaning temperature, and chemical concentration. A meter with poor internal geometry — sharp corners, blind bores, or dead zones behind the rotor end-plates — will not achieve the required turbulence and will fail a riboflavin test (a fluorescent tracer used to verify that cleaning solution reaches all internal surfaces).
SIP (Steam-in-Place) adds a thermal dimension: the meter must reach and maintain a minimum of 121 °C (at 15 psig steam pressure) for at least 30 minutes for the F₀ sterilisation concept to be satisfied in pharmaceutical applications. PD meters qualified for SIP must have housings, rotors, bearings, and seals that are thermally stable at these conditions — a standard that rules out some low-cost designs with polymer bearings rated only to 100 °C.
Potential for Residue Accumulation and Its Impact on Accuracy
Product residue accumulation in a PD meter does two things simultaneously: it poses a microbiological risk, and it degrades measurement accuracy. As deposits build up on the rotor surfaces or in the tight clearances of the measuring chamber, they reduce the effective chamber volume — causing the meter to over-read — and increase rotational resistance, which can cause the meter to stall at low flow rates. A tomato paste plant that did not validate its CIP program for a new PD meter installation found that after 90 days of operation, its batch weights had drifted 0.8% high — traced to a 0.3 mm caramelised tomato residue coating on the oval gear surfaces.
Validation Approaches for Sanitation Procedures
Regulatory validation of CIP and SIP procedures for metering equipment in pharmaceutical and food applications follows a tiered approach: Installation Qualification (IQ) — confirming the meter is installed per specification; Operational Qualification (OQ) — confirming it performs within stated accuracy at all operating conditions; Performance Qualification (PQ) — confirming it delivers the required sanitary outcome (i.e., microbial counts below specification) under production conditions. Many plant engineers focus heavily on IQ and OQ but treat PQ as a paperwork exercise. In a pharmaceutical context, a PQ that has never included actual microbiological swab results from the meter internals is not defensible under FDA or EMA inspection.
Calibration, Accuracy, and Repeatability
Internal Slippage (35%)
Temp/Viscosity Effect (25%)
Calibration Drift (20%)
Installation Effects (12%)
Electronics Noise (8%)
Based on field data aggregated from hygienic PD meter installations
Breakdown of major contributors to PD meter measurement uncertainty in hygienic process applications. Internal slippage and temperature/viscosity effects together account for 60% of the total uncertainty budget — both are addressable through proper calibration and temperature compensation.
Calibration Methods for PD Meters in Hygienic Lines
There are three practical calibration approaches for hygienic PD meters in service:
Water calibration with viscosity correction is the most common. The meter is calibrated on-bench or at a flow lab using water, and the K-factor (pulses per unit volume) is adjusted to account for the difference in slippage between water and the actual process fluid using a validated viscosity correction model. It is cost-effective but introduces model error if the process fluid’s viscosity is not precisely characterised.
Product calibration in a flow lab uses the actual process fluid at representative temperature and flow conditions. This is the most accurate approach and is mandatory for pharmaceutical applications where the meter is used in regulated batch records. The drawback is cost: sending a meter to a calibration laboratory with temperature-controlled product tanks and NIST-traceable gravimetric references adds significantly to commissioning cost but dramatically reduces measurement uncertainty.
In-situ verification by comparison uses a portable check meter (typically a Coriolis reference) installed temporarily in series with the installed PD meter. This is a practical tool for in-service drift monitoring without removing the meter from service — a significant advantage on production lines where downtime is measured in thousands of dollars per hour.
Factors Affecting Accuracy with High-Viscosity Fluids
As discussed in the viscosity section, higher viscosity generally improves PD meter accuracy by reducing slippage. However, several secondary effects can offset this benefit. First, at very high viscosities, the fluid’s resistance to flow through the clearances creates a pressure differential across the rotor that can introduce a small but systematic positive bias. Second, if the fluid is non-Newtonian (exhibiting shear-thinning behaviour, as most food emulsions, starch solutions, and yoghurt do), the effective viscosity in the tight clearances — where shear rates are highest — may be substantially lower than the bulk process viscosity, increasing slip in proportion. Specifying the meter’s performance with a rheological characterisation of the actual process fluid, not a single-point viscosity number, is best practice for non-Newtonian applications.
Best Practices for Regular Verification and Drift Tracking
Establishing a meter verification schedule based on process risk rather than arbitrary calendar intervals is an under-utilised approach that saves both cost and compliance headaches. A high-risk meter — one measuring a controlled active pharmaceutical ingredient in a batch record — should be verified at a frequency calibrated to its demonstrated drift rate over the first six months of service. A lower-risk meter metering a high-volume commodity ingredient with wide tolerance can be verified annually. Statistical process control (SPC) charts of meter K-factor over successive calibrations are a powerful tool: a trending K-factor signals seal wear or rotor erosion well before the meter goes out of specification.
Installation and Process Integration

Modern hygienic PD meters output 4–20 mA, HART, PROFIBUS PA, or Foundation Fieldbus signals — enabling direct integration into plant DCS and batch management systems without analog-to-digital conversion losses.
Piping Layout: Insertion vs. Inline Configurations
PD flowmeters are inherently inline devices — the entire flow stream passes through the measuring chamber. This distinguishes them from insertion-type meters (like hot-tap turbines or insertion ultrasonic probes) that sample only a portion of the flow profile. For hygienic applications, the inline configuration is actually advantageous: there is no partial-insertion geometry to create a dead zone, and the meter forms a closed, cleanable section of the pipeline.
The key installation requirement is upstream and downstream straight-run length. Unlike differential pressure meters that may need 20+ pipe diameters of straight run, most PD meters require only 5–10 diameters upstream and 3–5 downstream — a significant installation footprint saving in congested plant areas. In high-viscosity applications where flow velocities are inherently low and velocity profiles are fully developed, even these requirements are often relaxed by the manufacturer.
Mounting, Vibration, and Thermal Considerations
Hygienic PD meters should be installed with the rotor shaft horizontal wherever possible — gravity effects on the rotor can introduce bearing wear asymmetry in vertical shaft orientations over long service periods. Pipeline vibration from nearby pumps or compressors should be isolated with flexible connections, as cyclic mechanical stress on the meter housing can fatigue the rotor bearings and shift the calibration K-factor. In applications where the process fluid is hot (above 80 °C), thermal expansion of the meter housing must be accommodated by the piping supports — a rigid support structure that clamps both the meter and the pipe prevents differential thermal expansion and can create internal stress that distorts the rotor clearances, causing sudden accuracy shifts.
Signals, Wiring, and Data Integration with Control Systems
Modern hygienic PD meter transmitters support multiple output protocols. The dominant options in the food and pharmaceutical industries are:
- 4–20 mA analogue with HART superimposed — industry workhorse, supported by all PLCs and DCS platforms, allows secondary variable transmission (temperature, totalised volume) over the same loop
- PROFIBUS PA — digital fieldbus widely used in European pharmaceutical plants; enables full parameterisation and diagnostics from the control room without local access to the meter
- Foundation Fieldbus (FF H1) — preferred in large refinery and chemical complex applications; less common in hygienic sectors
- IO-Link / industrial Ethernet (PROFINET, EtherNet/IP) — emerging in new greenfield hygienic plants for higher data throughput and OPC UA integration with MES/ERP systems
Applications in Food, Pharmaceutical, and Biotech
Case Examples in Confectionery, Dairy, and Beverage Sectors
In confectionery manufacturing, glucose syrups, fondant masses, and chocolate coatings present some of the most demanding PD meter applications: viscosities ranging from 500 to 50,000 cP, temperatures spanning 30–80 °C, and high sugar content that caramelises if flow stops in a warm meter body. One leading confectionery manufacturer reported that after switching to a jacketed oval gear PD meter (with steam tracing on the meter housing to prevent product solidification during standby), their dosing CV (coefficient of variation) across a 12-month production period dropped from 1.2% to 0.18% — a figure that directly reduced chocolate overweight cost by approximately $240,000 per year on a single filling line.
In dairy processing, hygienic PD meters are deployed for cream dosing, cultured milk addition in stirred yoghurt, and cream cheese filling. The critical requirement here is accurate measurement at the low end of the turndown range — the dribble-rate addition of cream to a continuous stirred yoghurt mix may represent only 5–8% of the meter’s maximum flow range. A 20:1 turndown ratio with ±0.3% AR accuracy at the low end is the typical specification for cream dosing applications in a premium yoghurt plant.
In the beverage sector, syrup dosing in carbonated soft drinks (CSD) plants is a classic PD meter application. Syrup concentrations are up to 68° Brix (roughly 65–70% dissolved solids by weight), with viscosities between 200 and 800 cP. Accurate syrup dosing directly controls the finished beverage’s Brix and flavour intensity — a 0.5° Brix variance is at the edge of consumer-perceivable taste difference for many beverages. PD meters at ±0.2% AR accuracy easily achieve the ±0.1° Brix process control tolerance required in a well-run CSD plant.
Pharmaceutical and Biotech Slurry Handling Considerations
In pharmaceutical oral solid dosage (OSD) manufacturing, PD meters are used to meter binders, surfactants, and granulating agents into high-shear granulators. The fluid streams are typically non-Newtonian and can contain fine drug particles in suspension (slurries). The key challenges are: (1) preventing sedimentation in the meter body during process pauses; (2) avoiding shear damage to delicate active ingredients; and (3) ensuring that all wetted parts meet 21 CFR and ICH Q3D elemental impurity guidelines.
In bioprocessing, PD meters find application in media and buffer addition to bioreactors, where extreme accuracy at low flow rates matters. A 500-litre bioreactor receiving a micro-nutrient supplement at 0.5 L/h needs a meter accurate to within ±2% at that flow rate — a performance level achievable with a reciprocating piston PD meter but not with a standard electromagnetic or turbine meter that would be operating at the extreme low end of its range.
Regulatory Implications and Traceability Requirements
In both food and pharmaceutical sectors, the flow meter is increasingly part of the regulatory record. FDA 21 CFR Part 11 compliance for electronic batch records requires that the flow measurement data — including its calibration status, uncertainty, and audit trail — be preserved in a tamper-evident system. European GMP Annex 11 imposes similar requirements. This elevates the instrument specification conversation from “does the meter work accurately” to “can we prove the meter was working accurately when this batch was produced” — a requirement that has significant implications for calibration documentation, SPC data retention, and transmitter firmware version control.
Maintenance, Durability, and Life-Cycle Costs
Common Wear Mechanisms in PD Meters with Viscous Fluids
The primary wear mechanisms in hygienic PD meters are bearing wear (particularly in applications with abrasive particles), seal extrusion (from thermal cycling between process temperature and CIP/SIP temperature), and rotor surface erosion (from high-velocity particles in slurries or from the impingement of cleaning jets at excessive velocities). In purely viscous, particle-free applications — such as cream, glucose syrup, or pharmaceutical excipient solutions — bearing life of 5–10 years between overhauls is achievable. In applications with fine particles above 100 ppm by weight, annual bearing inspection is prudent.
Spare Parts Strategy and Service Intervals
A pragmatic spare parts strategy for hygienic PD meters in a production environment maintains at minimum: a complete set of dynamic seals (rated for the application temperature and CIP chemistry), a spare rotor assembly, and a spare set of bearings. With these components on the shelf, the MTTR (mean time to repair) following an unexpected failure is measured in hours rather than days — critical in a production environment where the cost of unplanned downtime may exceed $5,000 to $50,000 per hour depending on the line.
Total Cost of Ownership, Downtime, and Reliability Metrics
Purchase price comparisons between PD meters and alternative technologies are frequently misleading because they omit the largest components of lifecycle cost: maintenance labour, calibration, and unplanned downtime. The table below presents a representative 10-year TCO comparison for a 2-inch hygienic flow meter in a dairy processing application, based on publicly available cost benchmarks and operator experience data:
| Cost Category | Hygienic PD Meter (Oval Gear) | Coriolis Meter (2″) | Magnetic Flowmeter (2″) |
|---|---|---|---|
| Purchase / Installation (Year 0) | $4,500–$8,000 | $12,000–$20,000 | $3,000–$6,000 |
| Annual Calibration | $300–$600 | $400–$800 | $200–$400 |
| Seal Replacement (per event) | $80–$200 (every 2–3 years) | $150–$400 (every 5 years) | $100–$250 (every 4–5 years) |
| Bearing / Rotor Overhaul | $400–$800 (every 5–7 years) | None (no moving parts) | None (no moving parts) |
| Unplanned Downtime Events (10 yr) | 0.5–1 events avg. | 0.2–0.4 events avg. | 0.3–0.6 events avg. |
| Estimated 10-Year TCO | $8,000–$14,000 | $17,000–$28,000 | $6,500–$11,000 |
| Suitability for Viscous Fluids (>500 cP) | ✔ Excellent | ✔ Good (high cost) | ✘ Poor (low viscosity bias) |
| Suitability for Non-Conductive Fluids | ✔ Yes | ✔ Yes | ✘ No |
Estimates based on published industry benchmarks and operator cost data. Actual costs vary by geography, service contract terms, and specific product stream. Downtime cost not included (highly site-specific).
Selecting the Right PD Flowmeter: Decision Guide
Matching Flow Range, Viscosity, and Hygienic Requirements
The selection process for a hygienic high-viscosity PD meter is most reliably structured as a sequential filter: first eliminate technologies that cannot work (e.g., magnetic meters for non-conductive fluids), then narrow by hygienic certification requirement (3-A, EHEDG, or ASME BPE), then match accuracy and turndown to the process specification, then evaluate TCO, and finally assess vendor support and spare parts availability.
| Selection Criterion | Key Questions to Ask | Typical Requirement for Hygienic/High-Viscosity |
|---|---|---|
| Fluid conductivity | Is the fluid electrically conductive (>20 µS/cm)? | If NO → PD or Coriolis required |
| Viscosity range | Min / max viscosity across temperature range? | PD excels from 10 to 10,000+ cP |
| Flow range / turndown | Max : min flow ratio in operation? | Minimum 10:1; batching lines typically need 20:1+ |
| Accuracy requirement | Process tolerance (% AR) and regulatory basis? | ±0.2–0.5% AR for most hygienic applications |
| CIP / SIP compatibility | CIP chemical type, temperature, frequency? SIP required? | EPDM or FFKM seals; 316L wetted body; validated CIP program |
| Hygienic certification | Applicable standard (3-A, EHEDG, FDA, ASME BPE)? | Specify the standard in the purchase specification, not just “hygienic” |
| Particle content | Is the fluid clean or does it contain particles/fibers? | PD meters require particle-free fluids or upstream filtration |
| Pressure / temperature limits | Max operating pressure and temperature (process + CIP + SIP)? | Most hygienic PD meters: 16–25 bar; -20 to +150 °C |
| Output / integration | Required signal type and fieldbus protocol? | 4–20 mA / HART standard; PROFIBUS PA or IO-Link for digital plants |
| Pipe size | Nominal pipe diameter (DN)? | Most hygienic PD meters: DN10–DN100 (3/8″ to 4″) |
Evaluation Checklist for Vendor Qualifications and Support
- Does the supplier provide original certification documents (3-A symbol, EHEDG certificate, FDA material declarations) — not just claims on a datasheet?
- Can they supply a validated CIP/SIP protocol with traceable test data?
- Do they publish factory calibration certificates with NIST-traceable traceability for each shipped meter?
- What is the lead time for replacement rotors, seals, and bearings?
- Do they offer field service or can they recalibrate meters at the factory within a defined turnaround time?
- Are technical support engineers available who understand hygienic process applications — not just meter mechanics?
- Do they have documented installations in the same industry and application type as yours?
Teams working through this process will benefit from the broader instrument selection guidance available at Jade Ant Instruments’ flowmeter selection guide, which also covers comparison of PD meters with electromagnetic, turbine, and ultrasonic alternatives across diverse industry applications.
Risk Assessment and Long-Term Performance Expectations
A structured risk assessment for PD meter selection in a hygienic application should evaluate four risk dimensions: measurement risk (consequence of an out-of-spec reading — batch failure, product recall, regulatory action), hygiene risk (consequence of inadequate cleanability — contamination event, product recall), reliability risk (consequence of unexpected failure — production downtime cost, safety implications), and compliance risk (consequence of insufficient documentation — regulatory audit finding, loss of licence to operate). For most pharmaceutical applications, measurement and compliance risk dominate. For food processing lines, hygiene and reliability risk are typically the primary drivers. Mapping each application against this framework before specifying an instrument prevents the common mistake of over-engineering for one risk dimension while under-specifying for another.
Need a flow measurement expert to review your hygienic or high-viscosity application? Jade Ant Instruments specialises in precision flow instrumentation for demanding industrial environments. Our engineering team can assist with instrument selection, calibration review, and compliance documentation.
Selecting a positive displacement flowmeter for a hygienic or high-viscosity application is not a commodity procurement decision — it is an engineering judgement with regulatory, financial, and product quality consequences that extend years beyond the purchase date. The key decision factors we have explored in this guide are worth restating clearly.
From a technical standpoint, PD meters are uniquely well-suited to viscous, non-conductive, and low-velocity fluid streams — precisely the conditions where most competing technologies underperform. Their accuracy improves with viscosity (reducing slippage), they tolerate a wide turndown range, and their direct volumetric measurement principle requires no velocity profile assumptions. From a hygienic standpoint, what separates a compliant instrument from a risk is not which standard is cited in the brochure but whether the actual hardware — the rotor, the seals, the surface finish, the housing geometry — has been validated to meet that standard under real cleaning conditions with the actual CIP program used at your facility.
Validation and ongoing verification are not one-time events. The calibration K-factor drift of a PD meter over its service life is a running diagnostic of its mechanical health. An SPC chart of K-factor across successive calibration events is worth more for reliability management than any preventive maintenance schedule based on operating hours alone. Pilot testing — running a candidate meter on a representative side stream at production conditions for 30–60 days before full commitment — remains the most reliable way to validate that a specification on paper translates to performance in the real process environment.
For teams building the specification, Jade Ant Instruments’ comparison of flow measurement technologies and resources from authoritative industry bodies like 3-A SSI and EHEDG provide the technical foundation for defensible instrument selection decisions. Approach the process systematically, document your rationale, and the PD flowmeter you install will deliver the accuracy, hygiene, and longevity your process demands.